Coordination networks for sensing in medicine
| Consortium: | |
| Dr. Hana Bunzen University of Augsburg Chair of Solid State and Materials Chemistry, Institute of Physics | |
| Project: | Coordination networks for sensing in medicine |
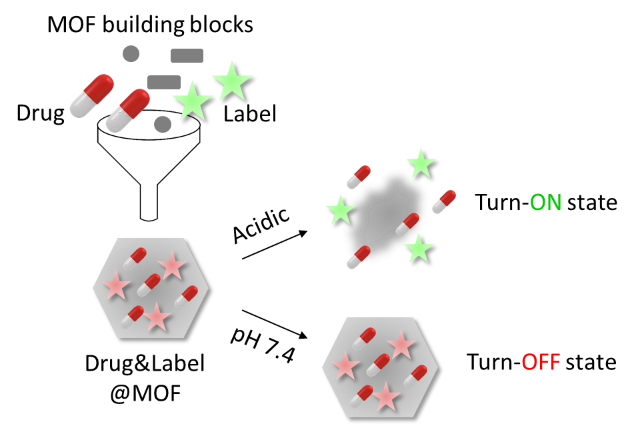
| Summary: | Functional smart materials, especially coordination networks, have recently attracted much attention in the field of biomedicine. In particular, utilizing stimuli-responsive MOF nanoparticles as drug delivery systems (DDSs) in cancer therapy have been widely investigated and were also of interest to us.(1-4) Drug delivery with nanoparticles is a complex process influenced by many factors. To analyze and optimize it, having a tool for efficient carrier tracking would be very helpful. Such tool would enable us to address many rudimentary but crucial issues in nanomedicine, including the nanoparticle distribution, monitoring and quantifying drug release, etc., which would further boost the field development. In our work we focus on developing trackable MOF-based nanocarriers for applications in cancer therapy. One of the greatest challenges in cancer therapy is to develop methods to deliver chemotherapy agents to tumor cells while reducing systemic toxicity to noncancerous cells. Compared with blood or healthy tissues, tumor microenvironments often have lower pH (as a consequence of the metabolism of cancer cells). Therefore, if suitable pH-sensitive smart materials are employed, localized drug release can be achieved. To monitor the drug release in situ, we suggest combining DDS with a label (e.g. a fluorophore or MRI-active species) which changes a measurable property upon its release from the DDS (as a consequence of the carrier decomposition in the acidic tumor microenvironment, Scheme 1). Such functional material is expected not only to be useful for (real-time) monitoring of the drug release, but also for selective labelling of cancer microenvironment, i.e. in enabling a differentiation between healthy (with no label released) and cancer microenvironment (with the label released). As an example of smart coordination network, we use pH-sensitive MOFs and combine them with selected labels to study host-guest interactions with regard to the turn-ON and turn-OFF properties. |
| Publications: | |
| R. Ettlinger, N. Moreno, N. Ziółkowska, A. Ullrich, H.-A. Krug von Nidda, D. Jirák, K. Kerl, H. Bunzen, “In Vitro Studies of Fe3O4‐ZIF‐8 Core–Shell Nanoparticles Designed as Potential Theragnostics” Part. & Part. Syst. Char. 2020, 2000185 DOI: 10.1002/ppsc.202000185 | |
| J. Schnabel, R. Ettlinger, H. Bunzen, “Zn‐MOF‐74 as pH‐Responsive Drug‐Delivery System of Arsenic Trioxide” ChemNanoMat 2020, 6, 1229-1236 DOI: 10.1002/cnma.202000221 | |
| R. Ettlinger, N. Moreno, D. Volkmer, K. Kerl, H. Bunzen, “Zeolitic Imidazolate Framework‐8 as pH‐Sensitive Nanocarrier for “Arsenic Trioxide” Drug Delivery” Chem. Eur J. 2019, 25, 13189-13196 DOI: 10.1002/chem.201902599 | |
| R. Ettlinger, M. Sönksen, M. Graf, N. Moreno , D. Denysenko, D. Volkmer, K. Kerl, H. Bunzen, “Metal–organic framework nanoparticles for arsenic trioxide drug delivery” J. Mater. Chem. B 2018, 6, 6481-6489 DOI: 10.1039/C8TB01899E |